Blood Antigens Markets
Universal Plasma :
Thanks to the availability of GlycoBAR low-cost antigen ligands, universal plasma will shortly become a reality.
Our blood group antigen ligands grafted on a cellulosic support (we have a proof of concept for this) are forming an affordable solution that can be used either in an individual plasma bag to make it universal or on plasma pools.

The universal plasma produced using GlycoBAR technology is equivalent to AB plasma, which hospitals are highly eager to use. There are many reasons for that:

Streamlined Inventory Management :
Hospitals and blood banks can maintain a single type of plasma, reducing the complexity and costs associated with storing multiple blood group plasmas.
Immediate Availability :
In emergencies, universal plasma can be readily available for transfusions without the need for blood type matching, speeding up treatment and potentially saving lives.
Reduced Risk of Transfusion Reactions :
By removing blood group antibodies, universal plasma minimizes the risk of hemolytic reactions, enhancing patient safety.
Increased Efficiency :
Simplifying the transfusion process allows healthcare providers to focus more on patient care and less on logistical challenges.
These improvements can lead to better resource allocation, reduced waste, and overall enhanced efficiency in medical facilities.
Universal Platelets :
The same benefits apply to platelets to an even greater extent.
Platelets are very short-lived blood components, lasting only a few days.
Being able to manage a single inventory of platelets would bring significant benefits to the health system.
Organ Transplants :
A recipient can accept only transplanted organs compatible with his ABO blood group otherwise his ABO antibodies will kill the transplanted organ.
As a result of the limited donors-recipient compatibility matrix, there is currently a huge shortage of available organs for transplant.
Blood group mismatch is a big issue and therefore providing a product allowing to resolve that issue is utmost important for saving lives.
Solution :
GlycoBAR partners develop extra corporal devices that filter the recipient blood so that blood group antibodies are eliminated from plasma.
Here is how it works :
The recipient patient’s blood is derived in an external circuitry where it is centrifugated with a plasmapheresis machine.
The plasma is purified from blood group antibodiesb thanks to a proprietary medical device using GlycoBAR ligands and all the blood components are then reinjected in the patient’s blood system.
After a few days of this treatment, the patient is ready to receive an organ of any blood group.
IVIG Purification :
The plasma fractionation industry producing so called IVIG products (Intra Venous Immunoglobulin type G) is facing a serious challenge:
As of today, for obvious logistic reasons and also because IgG blood group antibodies are not highly hemolytic, the plasma used in fractionation factories is not grouped (this is a mix of plasma of all groups).
Consequently, IVIG products contain isoagglutinins (the name for blood group antibodies in the plasma derived products).
While isoagglutinins are generally not highly hemolytic and typically safe for most patients, there have been occasional cases of hemolysis, especially in immunocompromised patients receiving higher doses of IVIG.
In 2015, the FDA raised concerns about this issue and encouraged IVIG suppliers to
remove isoagglutinins from their products.
However, due to the high cost and production challenges of blood group antigen ligands at the time, few suppliers complied with these recommendations.
There are two ways to remove isoagglutinins from IVIG :
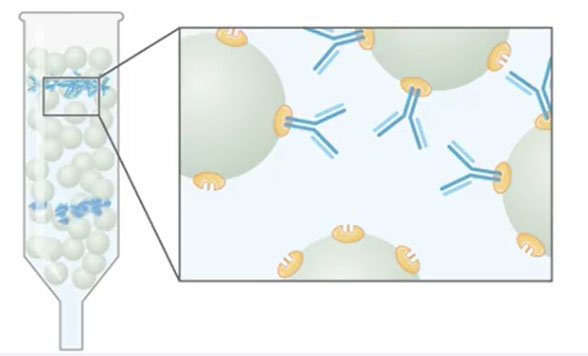
The first one is by using a traditional chromatographic column with a resin grafted with GlycoBAR ligands.
By adding a very simple and well-known affinity
purification step in their IVIG production process, IVIG suppliers can now eliminate completely isoagglutinins from their products and therefore provide a much safer solution to hospital for treating immune-deficient patients.
GlycoBAR has a partnership with a leading chromatographic resin supplier which is taking care of the grafting process and offer a turn-key solution to IVIG suppliers
Second one: